TREM2 on microglia cell surface binds to and forms functional binary complexes with heparan sulfate modified with 6-O-sulfation and iduronic acid
Authors:
Ilayda Ozsan McMillan, Li Liang, Guowei Su, Xuehong Song, Kelly Drago, Hua Yang, Claudia Alvarez, Amika Sood, James Gibson, Robert J. Woods, Chunyu Wang, Jian Liu, Fuming Zhang, Tom J. Brett, Lianchun Wang
Affiliation:
Division of Chemical Biology and Medicinal Chemistry, University of North Carolina at Chapel Hill, Eshelman School of Pharmacy, Chapel Hill, North Carolina, USA; Glycan Therapeutics, Raleigh, North Carolina, USA; Division of Pulmonary and Critical Care Medicine, Department of Internal Medicine, Washington University School of Medicine, St Louis, Missouri, USA; Department of Molecular Pharmacology and Physiology, University of South Florida Morsani College of Medicine, Tampa, Florida, USA; Department of Biological Sciences, Rensselaer Polytechnic Institute, Troy, New York, USA; Departments of Chemistry and Chemical Biology, Center for Biotechnology and Interdisciplinary Studies, Rensselaer Polytechnic Institute, Troy, New York, USA; Complex Carbohydrate Research Center, University of Georgia, Athens, Georgia, USA
Description:
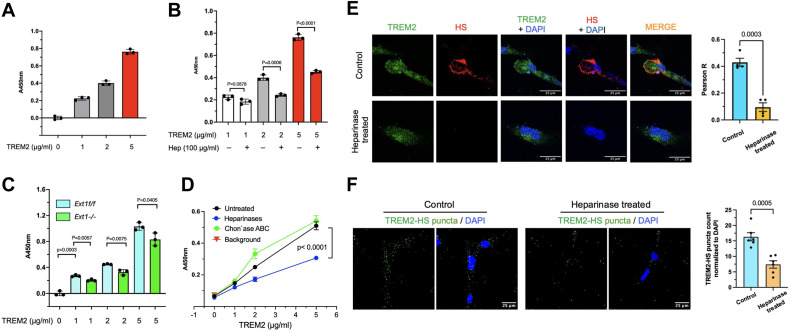
The triggering receptor expressed on myeloid cells-2 (TREM2), a pivotal innate immune receptor, orchestrates functions such as inflammatory responses, phagocytosis, cell survival, and neuroprotection. TREM2 variants R47H and R62H have been associated with Alzheimer's disease, yet the underlying mechanisms remain elusive. Our previous research established that TREM2 binds to heparan sulfate (HS) and variants R47H and R62H exhibit reduced affinity for HS. Building upon this groundwork, our current study delves into the interplay between TREM2 and HS and its impact on microglial function. We confirm TREM2's binding to cell surface HS and demonstrate that TREM2 interacts with HS, forming HS‑TREM2 binary complexes on microglia cell surfaces. Employing various biochemical techniques, including surface plasmon resonance, low molecular weight HS microarray screening, and serial HS mutant cell surface binding assays, we demonstrate TREM2's robust affinity for HS, and the effective binding requires a minimum HS size of approximately 10 saccharide units. Notably, TREM2 selectively binds specific HS structures, with 6‑O‑sulfation and, to a lesser extent, the iduronic acid residue playing crucial roles. N‑sulfation and 2O‑sulfation are dispensable for this interaction. Furthermore, we reveal that 6‑O‑sulfation is essential for HS‑TREM2 ternary complex formation on the microglial cell surface, and HS and its 6‑O‑sulfation are necessary for TREM2‑mediated ApoE3 uptake in microglia. By delineating the interaction between HS and TREM2 on the microglial cell surface and demonstrating its role in facilitating TREM2‑mediated ApoE uptake by microglia, our findings provide valuable insights that can inform targeted interventions for modulating microglial functions in Alzheimer's disease.
Publications:
- Ilayda Ozsan McMillan; Li Liang; Guowei Su; Xuehong Song; Kelly Drago; Hua Yang; Claudia Alvarez; Amika Sood; James Gibson; Robert J. Woods; Chunyu Wang; Jian Liu; Fuming Zhang; Tom J. Brett; Lianchun Wang; TREM2 on microglia cell surface binds to and forms functional binary complexes with heparan sulfate modified with 6-O-sulfation and iduronic acid; Journal of Biological Chemistry, 2024
Tags:
Glycosaminoglycans Heparan sulfate Peptides and proteins Surface plasmon resonanceRelated Chronicles:
Files:
| File Name | File Description | File Type | File Size | File URL |
|---|---|---|---|---|
| Supporting Information | Supporting Information | zip | 5.97 MB | Login to download |