Characterization of heparin interactions with Clostridioides difficile toxins and its potential as anti-CDI therapeutics
Authors:
Fuming Zhang, Shaohui Wang, Jiyuan Yang, Keith Fraser, James M. Gibson, Chunyu Wang, Jonathan S. Dordick, Anastasia Tomatsidou, Robert J. Linhardt, Lianchun Wang, Xingmin Sun
Affiliation:
Department of Chemical and Biological Engineering, Center for Biotechnology and Interdisciplinary Studies, Rensselaer Polytechnic Institute, Troy, NY 12180, USA; Department of Molecular Pharmacology and Physiology, Morsani College of Medicine, University of South Florida, Tampa, FL 33620, USA; Department of Biological Science, Center for Biotechnology and Interdisciplinary Studies, Rensselaer Polytechnic Institute, Troy, NY 12180, USA; Department of Molecular Medicine, Morsani College of Medicine, University of South Florida Tampa, FL 33620, USA
Description:
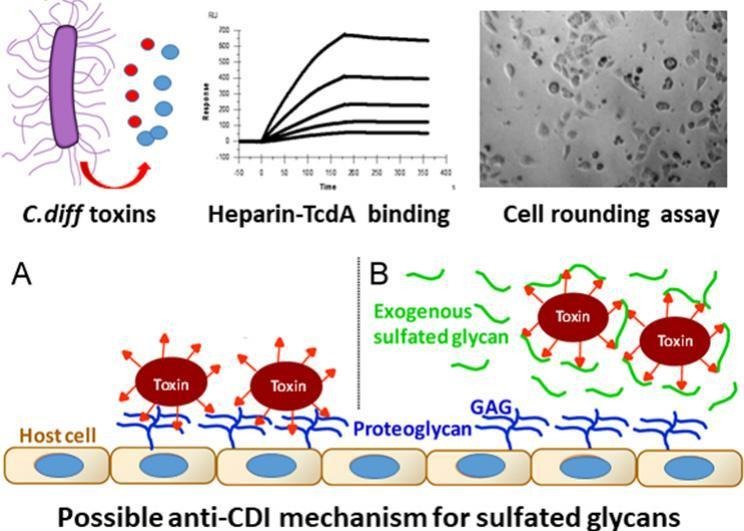
Clostridioides difficile ( C. difficile ) infection (CDI) is a life‑threatening healthcare‑associated infection occurring worldwide. C. difficile toxins (toxin A and toxin B) are the major virulence factors, causing CDI‑related diarrhea and complications. Recent studies have shown that sulfated glycosaminoglcans (GAGs) are involved in mediating the cellular entry of these toxins. Although interactions between GAGs and toxins were reported, their binding kinetics and the structure features of glycans that facilitate toxin interaction have not been thoroughly studied. This research utilized surface plasmon resonance (SPR) to directly measure the kinetics of interactions between heparin and various toxins. Both toxin A and toxin B bind to heparin with high affinity ( K D = 3.3 nM and 13.5 nM, respectively). SPR competition assay showed that both toxin A and B prefer binding to longer heparin chains and that all sulfation on the heparin chain is crucial for the heparin‑toxin interaction. Finally, an in vitro assay showed that heparin and non‑anticoagulant heparin inhibit the cell rounding caused by toxin A in HeLa cells.
Publications:
- Fuming Zhang; Shaohui Wang; Jiyuan Yang; Keith Fraser; James M. Gibson; Chunyu Wang; Jonathan S. Dordick; Anastasia Tomatsidou; Robert J. Linhardt; Lianchun Wang; Xingmin Sun; Characterization of heparin interactions with Clostridioides difficile toxins and its potential as anti-CDI therapeutics; Carbohydrate Polymers, 2025
Tags:
Glycosaminoglycans Heparin Peptides and proteins Surface plasmon resonanceRelated Chronicles:
Files:
No related files available