Are N-linked glycans intrinsically disordered?
Authors:
Eliza Gazaway, Rajan Kandel, Oliver C. Grant, Robert J. Woods
Affiliation:
Complex Carbohydrate Research Center and Department of Biochemistry and Molecular Biology, University of Georgia, 315 Riverbend Road, Athens, 30602, Georgia
Description:
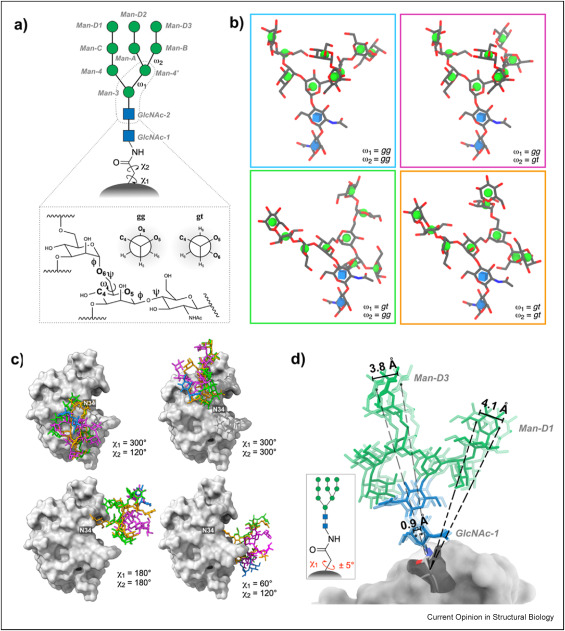
The covalent attachment of oligosaccharides to asparagine side chains on protein surfaces ( Nlinked glycosylation) is a ubiquitous modification that is critical to protein stability and function. Experimental 3D structures of glycoproteins in which the Nlinked glycans are well resolved are rare due to both the presumed flexibility of the Nlinked glycan and to glycan microheterogeneity. To surmount these limitations, computational modeling is often applied to glycoproteins, particularly to generate an ensemble of 3D shapes for the Nlinked glycans. While the number of glycoprotein modelling tools continues to expand, the available experimental data against which the predictions can be validated remains extremely limited. Here, we present our current understanding of the dynamic properties of Nlinked glycans, with a particular focus on features that impact their presentation (orientation) relative to the protein surface. Additionally, we review the limits of experimental and theoretical studies of glycoproteins, and ask the question, ' Are Nlinked glycans intrinsically disordered? '.
Publications:
- Eliza Gazaway; Rajan Kandel; Oliver C. Grant; Robert J. Woods; Are N-linked glycans intrinsically disordered?; Current Opinion in Structural Biology, 2025
Tags:
Atomistic simulation Glycosylation Modeling and Simulation OligosaccharidesRelated Chronicles:
Files:
No related files available